Fraunhofer IBMT Partner in the new EU Project “SafePolyMed”
Safer Drug Treatment and Enhanced Patient Empowerment
In the new EU project “SafePolyMed" an international research team sets out to provide physicians and pharmacists with innovative tools to increase drug treatment safety and educate patients on how to actively manage their own healthcare. The Fraunhofer Institute for Biomedical Engineering IBMT is contributing its many years of expertise in the field of health information systems to the new project.

Adverse drug reactions (ADRs) are a major burden to our healthcare and economic sys-tems. In Europe alone, approximately 197,000 deaths per year can be attributed to ADRs according to an assessment by the European Commission. The regular use of five or more medications concomitantly (polypharmacy), the coexistence of two or more long-term medical conditions or diseases (comorbidity) and genetic diversity have a major effect on drug efficacy and consequently, raise the incidence and severity of ADRs. Despite the fact that drug-drug interactions (DDIs) and drug-gene interactions (DGIs) are highly interconnected, in clinical practice, they are still considered separate entities. Hence, a more holistic approach taking into account individual disease states and drug-drug-gene interactions (DDGIs) is needed.
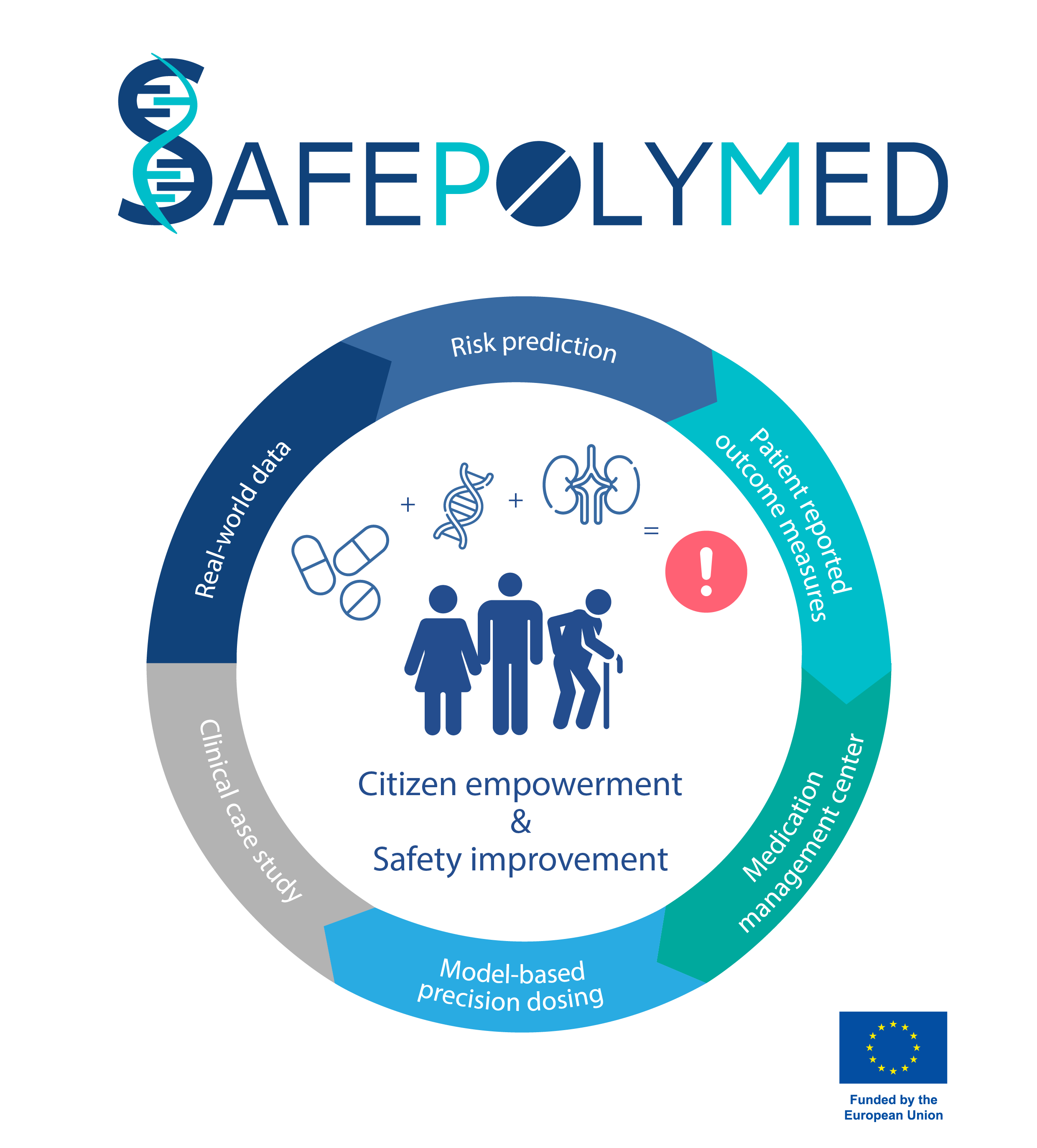
Aiming to increase overall patient safety, the new research project “SafePolyMed – Improving Safety in Polymedication by Managing Drug-Drug-Gene Interactions” seeks to develop innovative tools to define, assess and manage DDGIs for physicians and individual patients promoting enhanced patient education and empowerment. Bringing together eleven partner institutions from across Europe, “SafePolyMed” receives a total funding of 5.6 Mio Euro under the European Union’s “Horizon Europe” Framework Programme for Research and Innovation.
Developing a robust framework for improved ADR risk assessment
Distinct features and reliable measures to accurately assess individual risks of patients to experience ADRs are currently still missing. This is where "SafePolyMed" comes in: Over the next three and a half years the multidisciplinary research team strives to come up with a robust and well-defined risk score system that can assist physicians and pharmacists in assessing the individual risk of patients for ADRs and, thus, guide their decision-making process. Due to the high complexity of assessing real-life DDGI scenarios for different patient groups, providing these scientific evidence-based guidelines is extremely challenging and requires an integrated approach combining different tools, technologies, and perspectives.
“Within SafePolyMed, we will use machine learning and artificial intelligence (AI) techniques to analyse large real-world datasets gathering and integrating genomic information, demographic data, current and chronic health conditions and medication-related details of individual patients to gain a better understanding of individual patients’ risk for poor treatment outcomes and guide more personalised therapy decisions,” explains project coordinator Thorsten Lehr, Professor of Clinical Pharmacy at Saarland University, Germany. “Plus, medication-related safety is also strongly linked to dose regimens. Therefore, we will complement our analyses with mechanistic mathematical models to further assess complex drug interactions and calculate dose adaptation tailored to individual patients.”
In order to make those model-based dose recommendations more accessible and attractive for health care providers to include them in their daily routines, “SafePolyMed” will develop a prototype of an easy-to-use, web-based clinical decision support system. The benefits of all of the tools and applications developed within the project will be validated in a transnational “proof-of-principle” study.
Promoting active patient engagement for future drug safety and healthcare management
Alongside the clinical perspective, a major pillar of the project’s workplan is the active engagement of patients. According to Prof. Lehr: “A systematic documentation of the health status as perceived by the patients themselves by using specific questionnaires, so-called patient-reported outcome measures (PROMs), can help identify problems associated to ADRs early on. Thus, we will develop PROMs for assessing safety in drug therapy, particularly ADRs, throughout different patient groups.”
Fraunhofer Institute for Biomedical Engineering creates a central medication management platform
The Fraunhofer IBMT's mission is to create a medication management platform as a sustainable infrastructure that help foster patient involvement and bolster exchange with patient organisations to increase participation in healthcare and empower citizens and patients to take on a more active role in their own therapy management. Integrating different national patient-centered applications and modules, a comprehensive medication management center – interoperable within Europe’s health systems – will be set up, giving patients better, more standardized access to health-related information, which will enable them to adequately manage their own drug treatments. The knowledge and patient engagement hub will furthermore offer capacity building and training activities in close collaboration with European patient organisations.
Project Key Facts
Full Name: Improving Safety in Polymedication by Managing Drug-Drug-Gene Interactions (SafePolyMed)
Start Date: 1 June 2022
Duration: 42 months
Budget: 5.6 Mio €
Coordinator: Saarland University
Website: www.safepolymed.eu
Social Media: LinkedIn l Twitter
Project Partners
Germany
EURICE - European Research and Project Office GmbH
Fraunhofer Institute for Biomedical Engineering IBMT
LeukaNET
Saarland University
University Hospital of RWTH Aachen
Estonia
University of Tartu
Finland
University of Helsinki
Greece
Foundation for Research and Technology – Hellas
University of Patras
Slovenia
Gospodar zdravja d.o.o.
University of Ljubljana
The Netherlands
Leiden University Medical Center
Contact
Project Coordinator
Saarland University / Clinical Pharmacy
Prof. Thorsten Lehr
Phone: +49 681 302-70255
thorsten.lehr@mx.uni-saarland.de
Project Management
EURICE GmbH
Klara Schneider
Phone: +49 6894 388 1313
k.schneider@eurice.eu